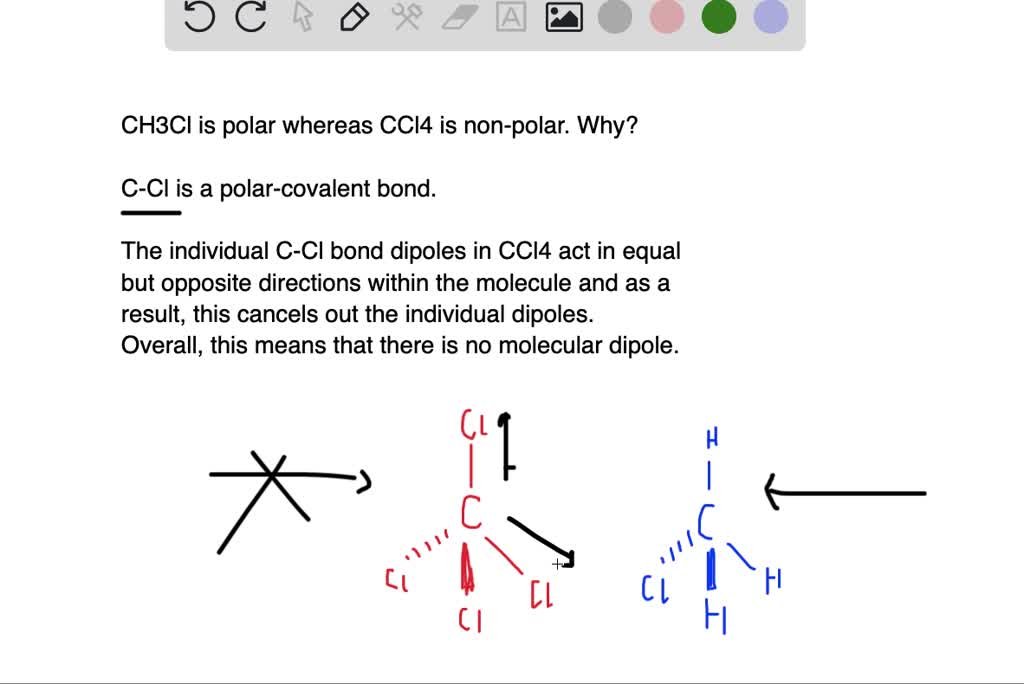
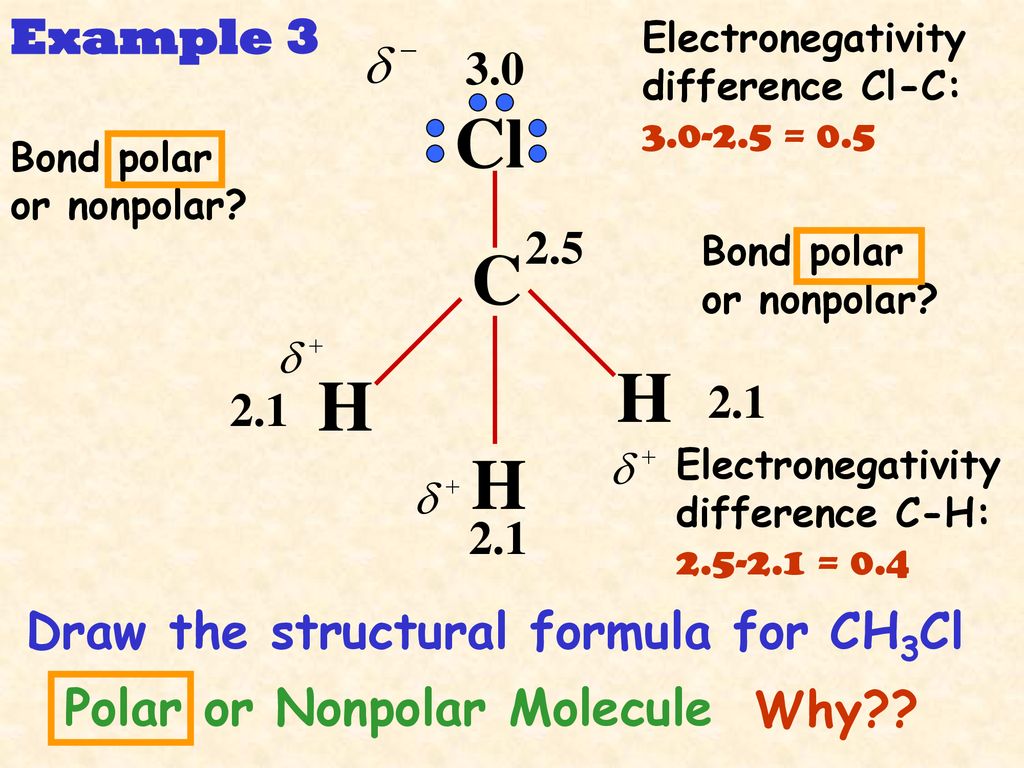
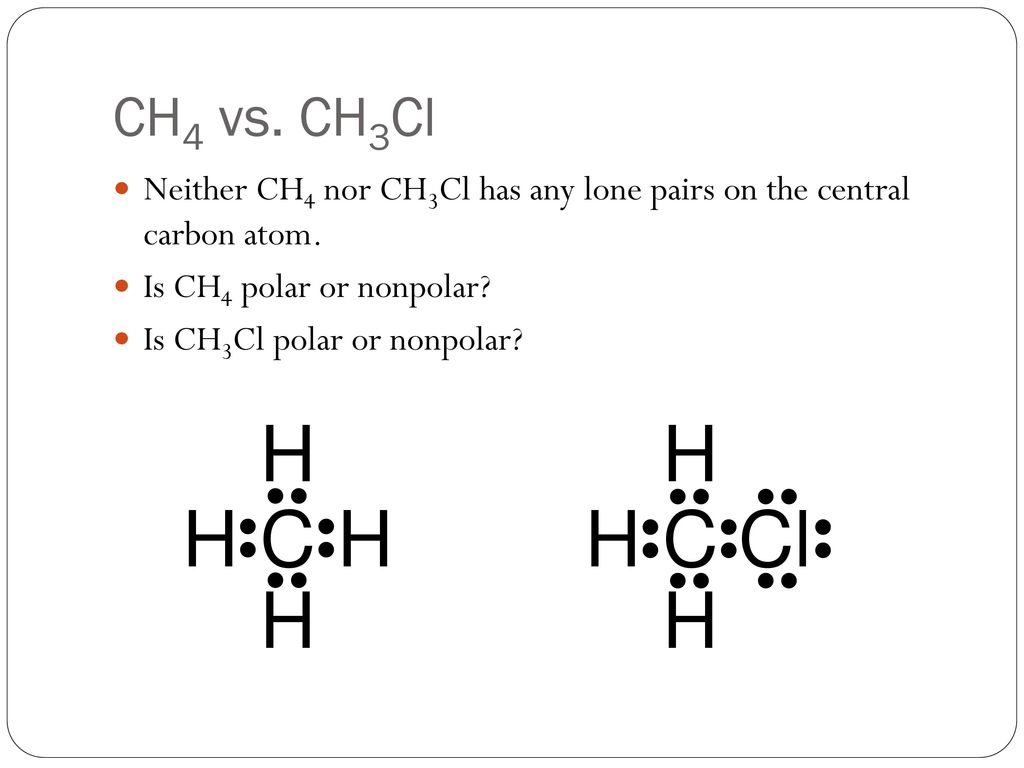
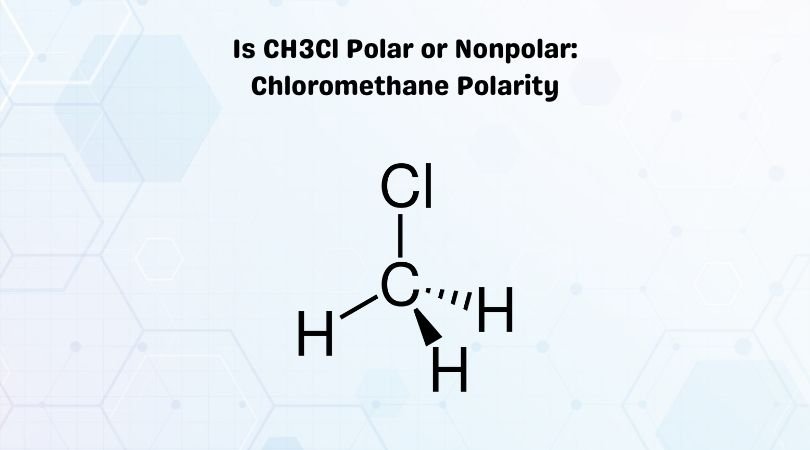
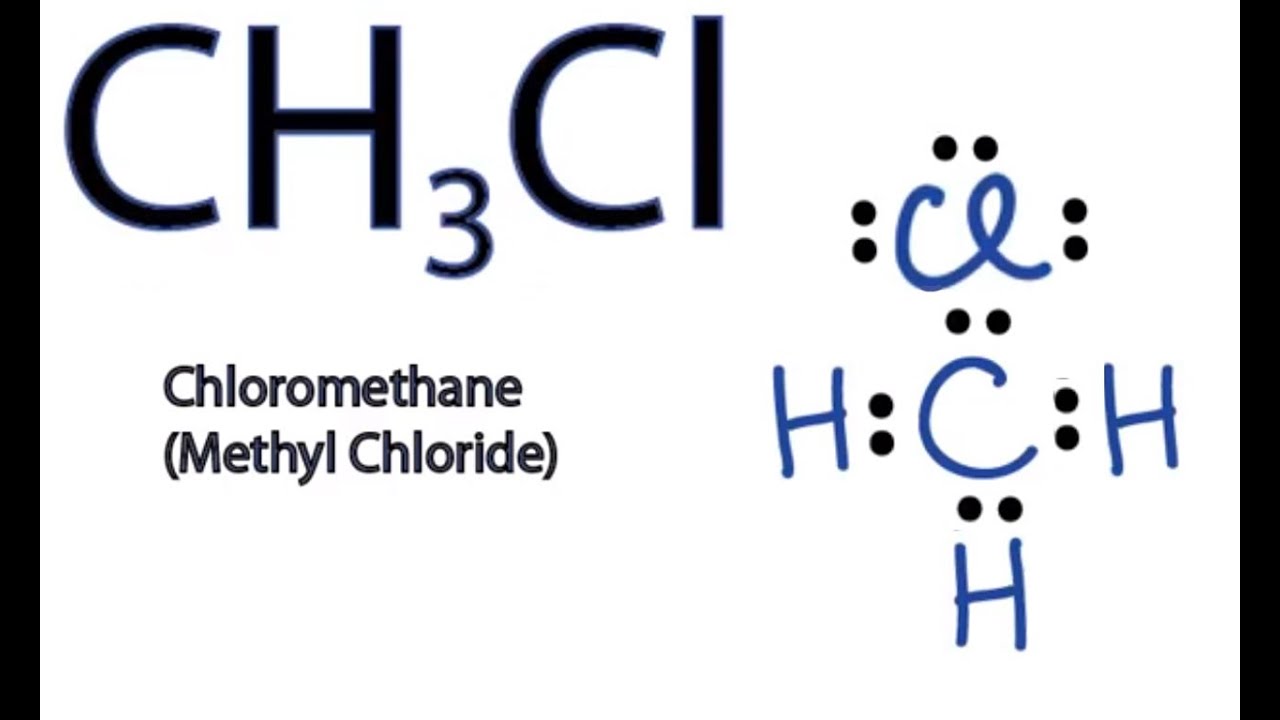Is the following molecule polar or nonpolar Give a reason for your answerElectronegativity values F 40 Cl 30Br 28 S 25 C 25 H21 The tetrahedral molecule chloromethane CH3Cl

Is CH3Cl Polar or Nonpolar? - Polarity of Chloromethane in 2023 | Molecular geometry, Polar, Molecular shapes

Is the molecule CH3Cl polar or nonpolar? If it is polar, specify the direction of its polarity. | Homework.Study.com

halides - Why does chloromethane have a larger dipole moment than chloroform? - Chemistry Stack Exchange