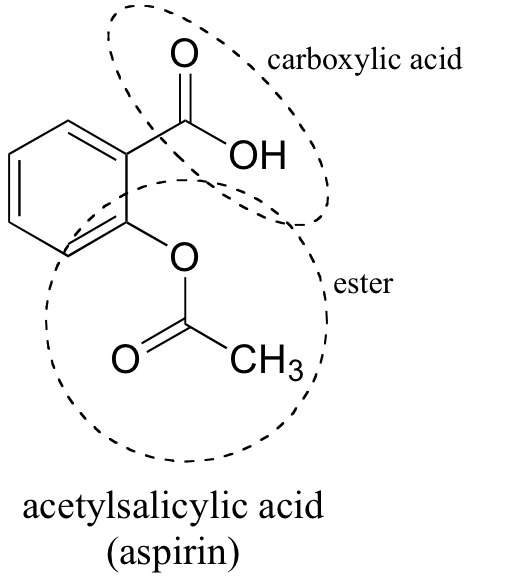
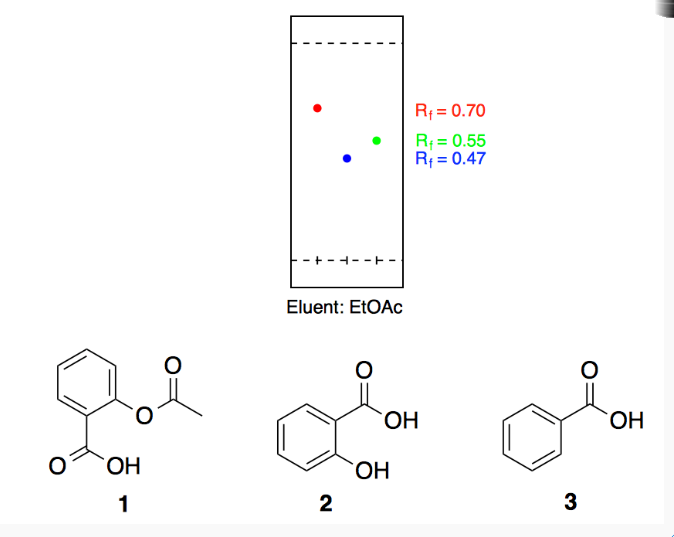
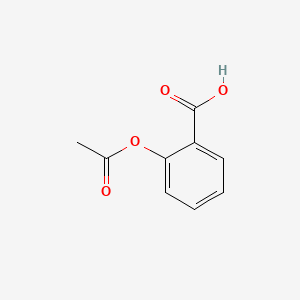
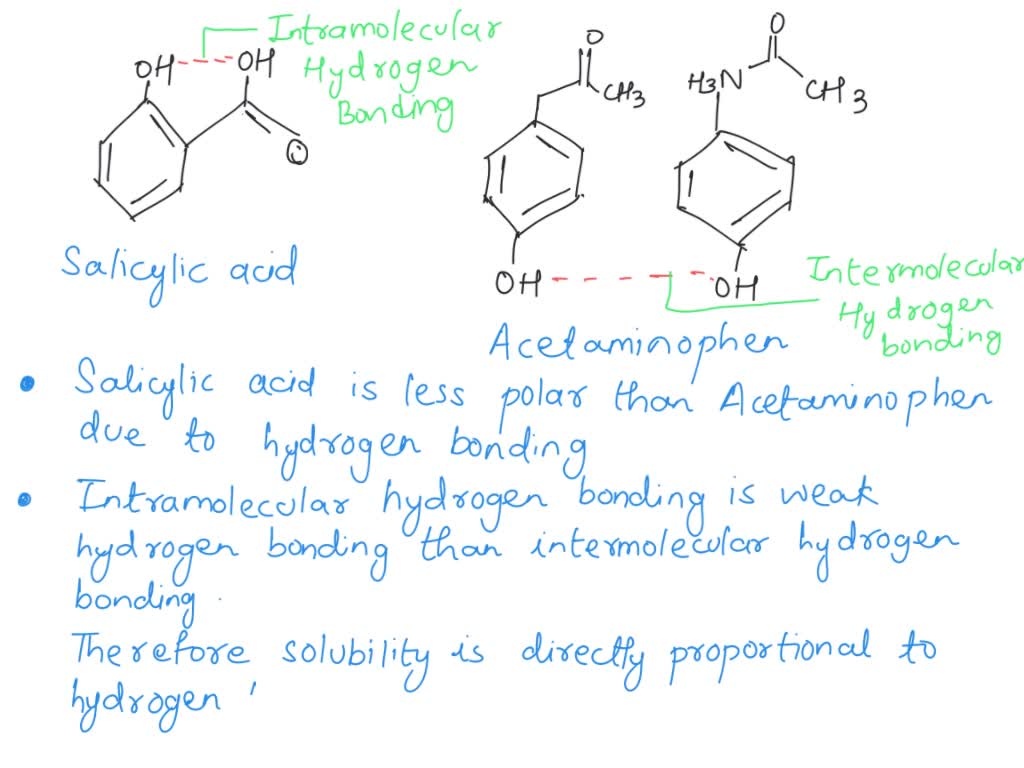
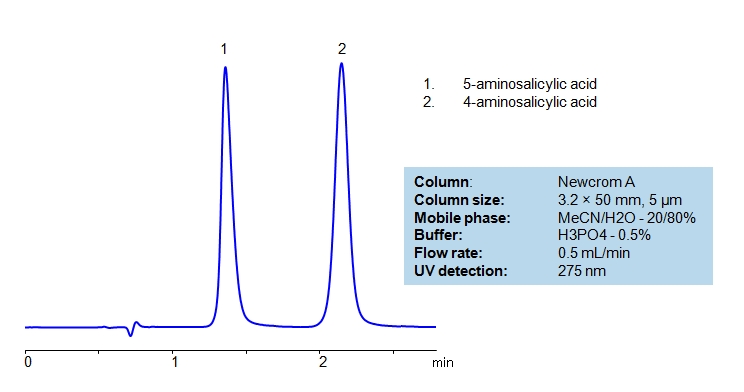
Between acetylsalicylic acid and benzoic acid, which is most polar if you look only at the structure (not at solubility in water g/l), and why? Why is that structure most polar? -
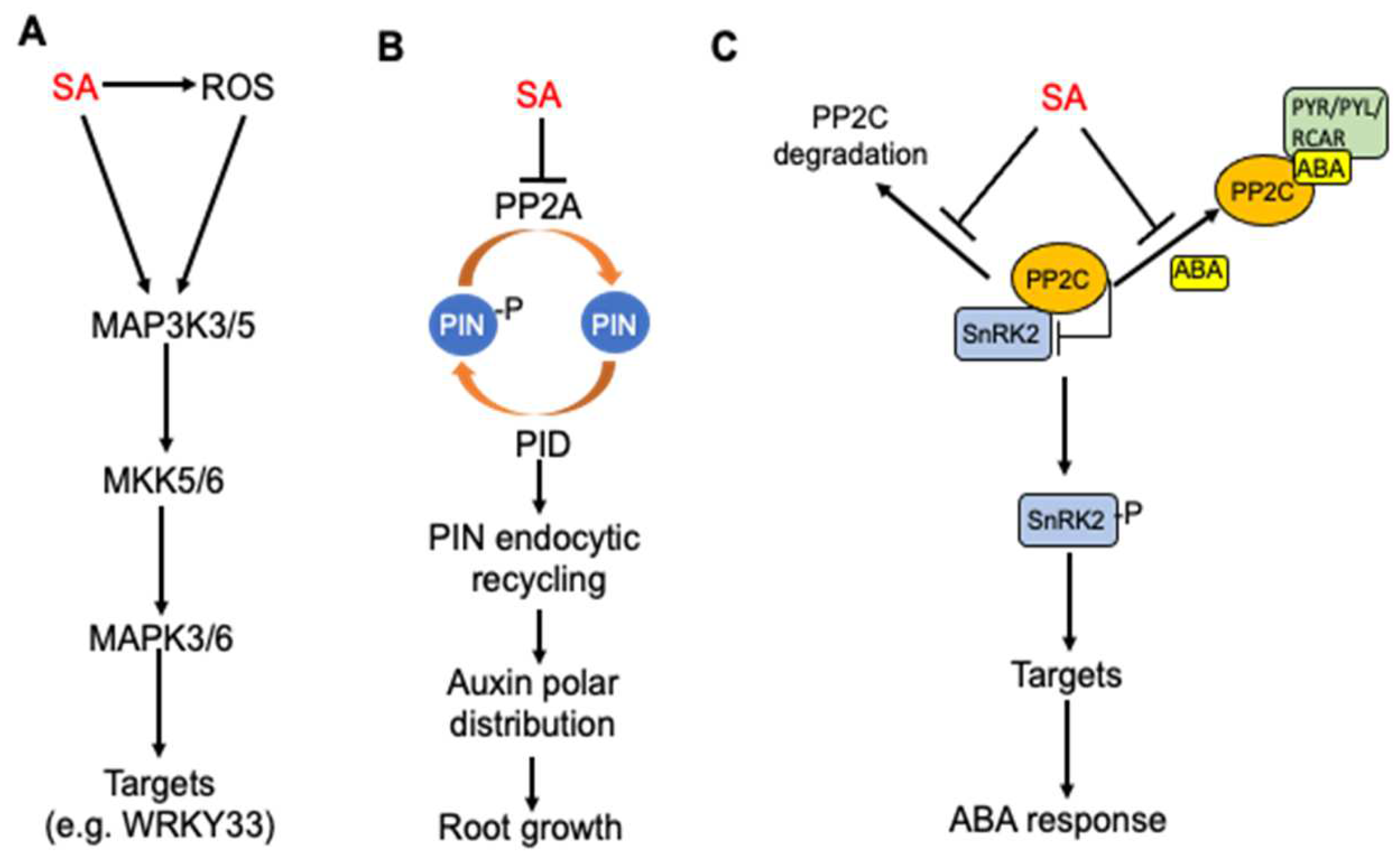
Cells | Free Full-Text | Shared and Related Molecular Targets and Actions of Salicylic Acid in Plants and Humans
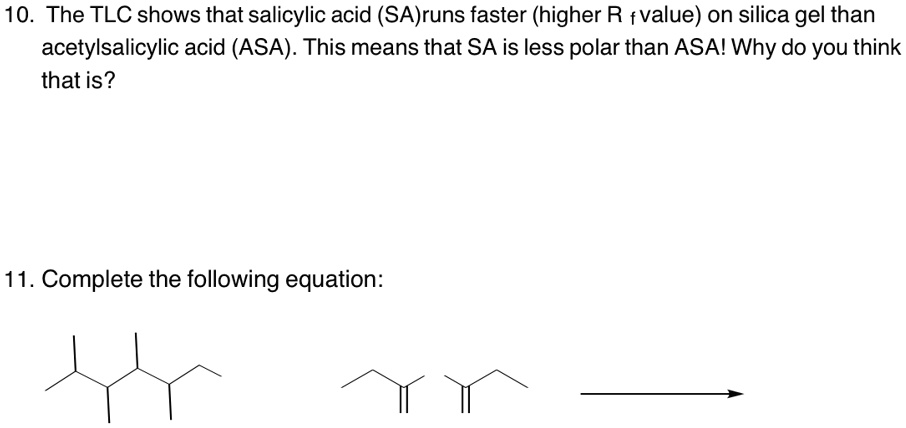
SOLVED: 10. The TLC shows that salicylic acid (SAJruns faster (higher R f value) on silica gel than acetylsalicylic acid (ASA): This means that SA is less polar than ASA! Why do

SOLVED: Salicylic acid reacts with iron(Ill) chloride to form a dark-colored precipitate; but acetylsalicylic acid does not react with iron(Ill) chloride. Why is this? a) Salicylic acid is more polar than acetylsalicylic

Salicylic acid, acetylsalicylic acid, methyl salicylate, salicylamide, and sodium salicylate in supercritical carbon dioxide: Solute – cosolvent hydrogen bonds formation - ScienceDirect